AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Keynote 4073/9/2023  
We compared therapeutic efficacy and AE between pembrolizumab ( N = 278) and atezolizumab ( N = 343) in combination with chemotherapy as the first-line treatment of advanced squamous NSCLC (Fig. Table 1 summarized study design, baseline characteristics and available endpoints of the trials in detail. 
A statistical test with P-value≤0.05 was considered as significant. HR 1 standed for pembrolizumab had longer PFS/OS or better ORR/less AE than atezolizumab incombination with chemotherapy. RR was evaluated similarly as above formulas. arm B (atezolizumab plus chemotherapy) was linked by arm C (chemotherapy), which was estimated by log HR AB = log HR AC − log HR BC, and its standard error (SE) for the log HR was. The log HR of the adjusted indirect comparison for arm A (pembrolizumab plus chemotherapy) vs. Based on the assumption that there is no significant treatment efficacy of carboplatin plus paclitaxel in comparison to carboplatin plus nab-paclitaxel for advanced squamous NSCLC, we calculated the adjusted indirect comparison using the following formulas as previously described. All of above data were derived from KEYNOTE-407 and IMpower131. Data of OS and PFS were extracted as hazard ratio (HR) and its 95% confidence interval (CI), while data of ORR and AE were extracted as risk ratio (RR) and its 95% CI. The clinical outcomes for our study were overall survival (OS), progression-free survival (PFS), objective response rate (ORR) and adverse event (AE). Therefore, we performed an indirect comparison of KEYNOTE-407 and IMpower131 to explore the optimal choice of anti-PD-(L)1 treatment for advanced squamous NSCLC in combination with chemotherapy. However, there is no head-to-head comparison of pembrolizumab plus chemotherapy vs. Meanwhile, another randomized phase III study (IMpower131) showed that combined carboplatin plus nab-paclitaxel with atezolizumab, an anti-programmed death ligand 1 (anti-PD-L1) antibody, also improved the therapeutic efficacy for those patients. Recently, a randomized phase III trial (KEYNOTE-407) reported that adding pembrolizumab, an anti-programmed death 1 (anti-PD-1) antibody, in combination with carboplatin plus paclitaxel/nab-paclitaxel decreases the mortality risk for advanced squamous NSCLC. Therefore, we still need to explore a better therapeutic regimen for advanced squamous NSCLC. The standard treatment for advanced squamous NSCLC includes platinum-based doublet chemotherapy, such as taxane-platinum combination, which has poor efficacy. However, this advancement has not been achieved in squamous NSCLC given the lack of efficacy and there are currently no approved targeted agents for squamous NSCLC. Patients with lung adenocarcinoma whose tumor harbor specific gene mutations, such as epidermal growth factor receptor (EGFR) mutation or anaplastic lymphoma kinase (ALK) fusion, derive significant benefit from targeted agents, tyrosine kinase inhibitors (TKIs), and have better prognosis. Yaxiong Zhang and Huaqiang Zhou contributed equally to this work.Īdenocarcinoma and squamous carcinoma are two most common histological subtype of advanced non-small-cell lung cancer (NSCLC). 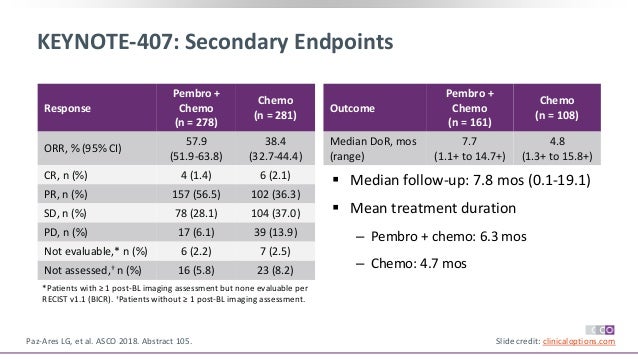
Our analysis raises the hypothesis that anti-PD-1 antibody therapy in combination with chemotherapy may have superior efficacy compared to anti-PD-L1 antibody combination for patients with PD-L1 low/negative advanced squamous NSCLC. For PD-L1 high patients, pembrolizumab and atezolizumab showed similar OS and PFS. For overall patients, pembrolizumab had significantly superior OS (hazard ratio (HR) with 95% confidence interval, 0.67, 0.47–0.94 P = 0.02) and numerically better PFS (HR, 0.79, 0.60–1.04 P = 0.10) than atezolizumab, while they had similar ORR, all cause AE and grade 3–5 AE. The clinical outcomes were overall survival (OS), progression-free survival (PFS), objective response rate (ORR) and adverse event (AE). Therefore, we performed an indirect comparison to explore the optimal choice of anti-PD-(L)1 treatment for advanced squamous NSCLC in combination with chemotherapy. atezolizumab (anti-PD-L1) plus chemotherapy. However, there is no head-to-head comparison of pembrolizumab (anti-PD-1) plus chemotherapy vs. Recent randomized phase III trials (KEYNOTE-407 and IMpower131) reported that adding anti-programmed death (ligand) 1 (anti-PD-(L)1) antibodies in combination with taxane-platinum improve the therapeutic efficacy for advanced squamous non-small-cell lung cancer (NSCLC). 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
